39. Synthesis and Reactivity of Ruthenium(BINAP)(PPh3)
Angew. Chem. Int. Ed. 2024, 63, e202318684
Y. Zhou, N. H. Wensink, A.-F. Pécharman, F. M. Miloserdov*
Hot Paper

38. Rim-Differentiated Pillar[5]arene-Modified Surfaces for
Rapid PFOA/PFOS Detection
Chem. Commun. 2024, Accepted Manuscript
T.-N. Gao, Z. Yang, J. M. S. Goed, H. Zuilhof,* F. M. Miloserdov*
37. 2-Methylimidazole-1-(N–tert-octyl)sulfonimidoyl Fluoride: A Bench-Stable Alternative to SOF4 as Precursor to N,O-Substituted S(VI) Compounds
Angew. Chem. Int. Ed. 2024, e202406915
N. Lional, F. M. Miloserdov*, H. Zuilhof*

36. 2D and 3D Self-Assembly of Fluorine-Free Pillar-[5]-Arenes and Perfluorinated Diacids at All-Aqueous Interfaces
Adv. Sci. 2024, 2401807
L. W. Honaker, T.-N. Gao, K. R. de Graaf, T. V. M. Bogaardt, P. Vink, T. Stürzer, G. Kociok-Köhn, H. Zuilhof,* F. M. Miloserdov,* S. Deshpande*

35. Rim-Based Binding of Perfluorinated Acids to Pillararenes Purifies Water
Angew. Chem. Int. Ed. 2024, 63, e20240347
T.-N. Gao, S. Huang, R. Nooijen, Y. Zhu, G. Kociok-Köhn, T. Stürzer, G. Li, J. H. Bitter, G. I. J. Salentijn, B. Chen, F. M. Miloserdov,* H. Zuilhof*

34. A Platform for the Liebeskind–Srogl Coupling of Heteroaromatic Thioethers for Medicinal-Chemistry-Relevant Transformations
Org. Lett. 2024, 26, 2852
V. Koolma, R. Staiger, M. Schühle, A. Bixenmann, E. Bauschatz, M. Schmid, F. M. Miloserdov,* B. Herlé*

33. Synthesis of Large Macrocycles with Chiral Sulfur Centers via Enantiospecific SuFEx and SuPhenEx Click Reactions
J. Org. Chem. 2023, 88, 15658
Y. Chao, M. Subramaniam, N. Kayambu, Y. Zhu, V. Koolma, Z. Hao, S. Li, Y. Wang, I. Hudoynazarov, F. M. Miloserdov, H. Zuilhof*

32. Enantiospecific Synthesis of Aniline-Derived Sulfonimidamides
Org. Lett. 2023, 25, 5666
D.-D. Liang, N. Lional, B. Scheepmaker, M. Subramaniam, G. Li, F. M. Miloserdov,* H. Zuilhof*

31. Isolation of Bis- and Mono-Cyclometallated Ru−IMes Complexes upon Reaction of [Ru(PPh3)3HCl], IMes and ZnMe2
Eur. J. Inorg. Chem. 2023, 26, e202300037
A.-F. Pecharman, E. M. Roberts, F. M. Miloserdov, V. Varela-Izquierdo, M. F. Mahon, M. K. Whittlesey*

30. Sulfur–Phenolate Exchange: SuFEx-Derived Dynamic Covalent Reactions and Degradation of SuFEx Polymers
Angew. Chem. Int. Ed. 2022, 61, e202207456
Y. Chao, A. Krishna, M. Subramaniam, D.-D. Liang, S. P. Pujari, A. C.-H. Sue, G. Li, F. M. Miloserdov, H. Zuilhof*

29. Experimental and Computational Studies of Ruthenium Complexes Bearing Z‑Acceptor Aluminum-Based Phosphine Pincer Ligands
Inorg. Chem. 2022, 61, 20690
C. J. Isaac, C. I. Wilson, A. L. Burnage, F. M. Miloserdov, M. F. Mahon, S. A. Macgregor*, M. K. Whittlesey*

Binding S(VI) to alkynes
Nat. Synth. 2022, 1, 415 (News & Views Article, non-refereed)
F. M. Miloserdov, H. Zuilhof*
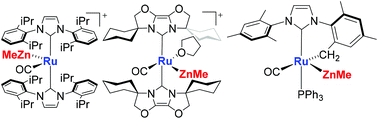
28. Zinc-Promoted ZnMe/ZnPh Exchange in Eight-Coordinate [Ru(PPh3)2(ZnMe)4H2]
Angew. Chem. Int. Ed. 2022, 61, e202117495
L. Sotorrios, F. M. Miloserdov,* A.-F. Pécharman, J. P. Lowe, S. A. Macgregor,* M. F. Mahon, M. K. Whittlesey*

27. Structure and Reactivity of [Ru-Al] and [Ru-Sn] Heterobimetallic PPh3-Based Complexes
Organometallics 2022, 41, 2716
C. J. Isaac, F. M. Miloserdov,* A.-F. Pécharman, J. P. Lowe, C. L. McMullin,* M. K. Whittlesey*

26. Bonding and Reactivity of a Pair of Neutral and Cationic Heterobimetallic RuZn2 Complexes
Inorg. Chem. 2021, 60, 16256
F. M. Miloserdov,* A.-F. Pécharman, L. Sotorrios, N. A. Rajabi, J. P. Lowe, S. A. Macgregor,* M. F. Mahon, M. K. Whittlesey*

Publications prior to WUR
25. Impact of the Novel Z-Acceptor Ligand Bis{(ortho-diphenylphosphino)phenyl}zinc (ZnPhos) on the Formation and Reactivity of Low-Coordinate Ru(0) Centers
Inorg. Chem. 2020, 59, 15606
F. M. Miloserdov,* C. J. Isaac, M. L. Beck, A. L. Burnage, J. C. B. Farmer, S. A. Macgregor,* M. F. Mahon, M. K. Whittlesey*

24. Zn-Promoted C-H Reductive Elimination and H2 Activation via a Dual Unsaturated Heterobimetallic Ru-Zn Intermediate
J. Am. Chem. Soc. 2020, 142, 6340
F. M. Miloserdov,* N. A. Rajabi, J. P. Lowe, M. F. Mahon, S. A. Macgregor,* M. K. Whittlesey*

23. Transforming PPh3 into bidentate phosphine ligands at Ru–Zn heterobimetallic complexes
Dalton Trans. 2019, 48, 14000
N. O’Leary, F. M. Miloserdov,* M. Mahon,* M. K. Whittlesey*
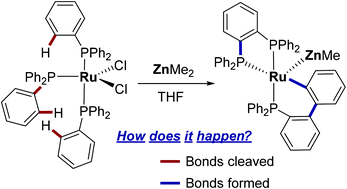
22. Heterobimetallic Ruthenium-Zinc Complexes with Bulky N-Heterocyclic Carbenes: Syntheses, Structures and Reactivity
Dalton Trans. 2019, 48, 4176
M. Espinal-Viguri, V. Varela-Izquierdo, F. M. Miloserdov, I. M. Riddlestone, M. F. Mahon*, M. K. Whittlesey*
21. Collected Mass Spectrometry Data on Monoterpene Indole Alkaloids from Natural Product Chemistry Research
Scientific Data 2019, 6, 15
A. E. Fox Ramos, [30 authors including FMM], M. A. Beniddir*
20. Unified Total Synthesis of Pyrroloazocine Indole Alkaloids Sheds Light on Their Biosynthetic Relationship
J. Am. Chem. Soc. 2018, 140, 5393
F. M. Miloserdov, M. S. Kirillova, M. E. Muratore, A. M. Echavarren*

19. Total Syntheses of Pyrroloazocine Indole Alkaloids: Challenges and Reaction Discovery (Review)
Org. Chem. Front. 2018, 5, 273
M. S. Kirillova, F. M. Miloserdov, A. M. Echavarren*

Hydroarylation of Alkynes using Cu, Ag and Au Catalysts
Catalytic Hydroarylation of Carbon-Carbon Multiple Bonds
Eds. L. Ackermann, T. B. Gunnoe, L. G. Habgood, Wiley-VCH, 2018
M. S. Kirillova, F. M. Miloserdov, A. M. Echavarren
18. On the Structure of [K(crypt-222)]+CF3–
Helv. Chim. Acta 2018, 101, e1800015
R. L. Harlow,* J. Benet-Buchholz,* F. M. Miloserdov, A. I. Konovalov, W. J. Marshall, E. C. Escudero-Adán, E. Martin, A. Lishchynskyi, V. V. Grushin*
17. The Trifluoromethyl Anion: Evidence for [K(crypt-222)]+CF3–
Helv. Chim. Acta 2017, 100, e1700032
F. M. Miloserdov, A. I. Konovalov, E. Martin, J. Benet-Buchholz, E. C. Escudero-Adán, A. Lishchynskyi, V. V. Grushin*
16. The Trifluoromethyl Anion
Angew. Chem. Int. Ed. 2015, 54, 15289
A. Lishchynskyi, F. M. Miloserdov, E. Martin, J. Benet-Buchholz, E. C. Escudero-Adán, A. I. Konovalov, V. V. Grushin*

15. Ruthenium-Catalyzed Nucleophilic Fluorination of Halobenzenes
Chem. Commun. 2015, 51, 13527
A. I. Konovalov, E. O. Gorbacheva, F. M. Miloserdov, V. V. Grushin*

14. Exceedingly Facile Ph–X Activation (X=Cl, Br, I) with Ruthenium(II): Arresting Kinetics, Autocatalysis, and Mechanisms
Angew. Chem. Int. Ed. 2015, 54, 8466
F. M. Miloserdov, D. McKay, B. K. Muñoz, H. Samouei, S. A. Macgregor*, V. V. Grushin*

13. Well-Defined CuC2F5 Complexes and Pentafluoroethylation of Acid Chlorides
Angew. Chem. Int. Ed. 2015, 54, 5218
L. I. Panferova, F. M. Miloserdov, A. Lishchynskyi, M. Martínez Belmonte, J. Benet-Buchholz, V. V. Grushin*

12. Solid-State Structure and Solution Reactivity of [(Ph3P)4Ru(H)2] and Related Ru(II) Complexes Used in Catalysis: A Reinvestigation
Organometallics 2014, 33, 7279
H. Samouei, F. M. Miloserdov, E. C. Escudero-Adán, V. V. Grushin*

11. On the Feasibility of Nickel-Catalyzed Trifluoromethylation of Aryl Halides
Organometallics 2014, 33, 6531
J. Jover, F. M. Miloserdov, J. Benet-Buchholz, V. V. Grushin* and F. Maseras*

10. Alcoholysis of Fluoroform
J. Fluorine Chem. 2014, 167, 105
F. M. Miloserdov, V. V. Grushin*

9. The Challenge of Palladium-Catalyzed Aromatic Azidocarbonylation: From Mechanistic and Catalyst Deactivation Studies to a Highly Efficient Process
Organometallics 2014, 33, 736
F. M. Miloserdov, C. L. McMullin, M. Martínez Belmonte, J. Benet-Buchholz, V. I. Bakhmutov, S. A. Macgregor*, V. V. Grushin*;

8. Palladium-Catalyzed Aromatic Azidocarbonylation
Angew. Chem., Int. Ed. 2012, 51, 3668
F. M. Miloserdov, V. V. Grushin*

7. CF3–Ph Reductive Elimination from [(Xantphos)Pd(CF3)(Ph)]
Organometallics 2012, 31, 1315
V. I. Bakhmutov,* F. Bozoglian, K. Gómez, G. González, V. V. Grushin,* S. A. Macgregor,* E. Martin, F. M. Miloserdov, M. A. Novikov, J. A. Panetier, L. V. Romashov

6. Formation of Binuclear Pyrazolate-bridged Palladium Carboxylates in the Reactions of [Pd(Hdmpz)4](OOCR)2 (R = Me, t-Bu, Ph) with Heterometallic Pd-Co Acetate
Russ. J. Inorg. Chem. 2009, 54, 1590
E. V. Perova, F. M. Miloserdov, M. Ya. Yakovleva, I. P. Stolyarov, S. E. Nefedov*
5. Effect of the Nature of Carboxylate Anion on the Features of Intramolecular Hydrogen Bonding in [Pd(Hdmpz)4](OOCR)2 (R = Me, t-Bu, Ph)
Russ. J. Inorg. Chem. 2009, 54, 1378
E. V. Perova, F. M. Miloserdov, M. Ya. Yakovleva, I. P. Stolyarov, S. E. Nefedov*
4. The Electrocatalytic Multicomponent Assemling of Isatins, 3-Methyl-2-pyrazolin-5-ones and Malononitrile: Facile and Convenient Way to Functionalized Spirocyclic[indole-3,4´-pyrano[2,3-c]pyrazole] System
Mol. Divers. 2009, 13, 47
M. N. Elinson,* A. S. Dorofeev,* F. M. Miloserdov, G. I. Nikishin*
3. Catalysis of Salicylaldehydes and Two Different C–H Acids with Electricity: First Example of an Efficient Multicomponent Approach to the Design of Functionalized Medicinally Privileged 2-Amino-4H-Chromene Scaffold
Adv. Synth. Catal. 2008, 350, 591
M. N. Elinson,* A. S. Dorofeev,* F. M. Miloserdov, A. I. Ilovaisky, S. K. Feducovich, P. A. Belyakov, G. I. Nikishin

2. Electrocatalytic Multicomponent Transformation of Cyclic 1,3-Diketones, Isatins, and Malononitrile: Facile and Convenient Way to Functionalized Spirocyclic (5,6,7,8-Tetrahydro-4H-Chromene)-4,3´-Oxindole System
Tetrahedron 2007, 63, 10543
M. N. Elinson,* A. I. Ilovaisky, A. S. Dorofeev,* V. M. Merkulova, N. O. Stepanov, F. M. Miloserdov, Y. N. Ogibin, G. I. Nikishin

1. The Implication of Electrocatalysis in MCR Strategy: Electrocatalytic Multicomponent Transformation of Cyclic 1,3-Diketones, Aldehydes and Malononitrile into Substituted 5,6,7,8-Tetrahydro-4H-Chromenes
Eur. J. Org. Chem. 2006, 4335
M. N. Elinson,* A. S. Dorofeev, S. K. Feducovich, S. V. Gorbunov, R. F. Nasybullin, F. M. Miloserdov, G. I. Nikishin
